REDEEM Safety
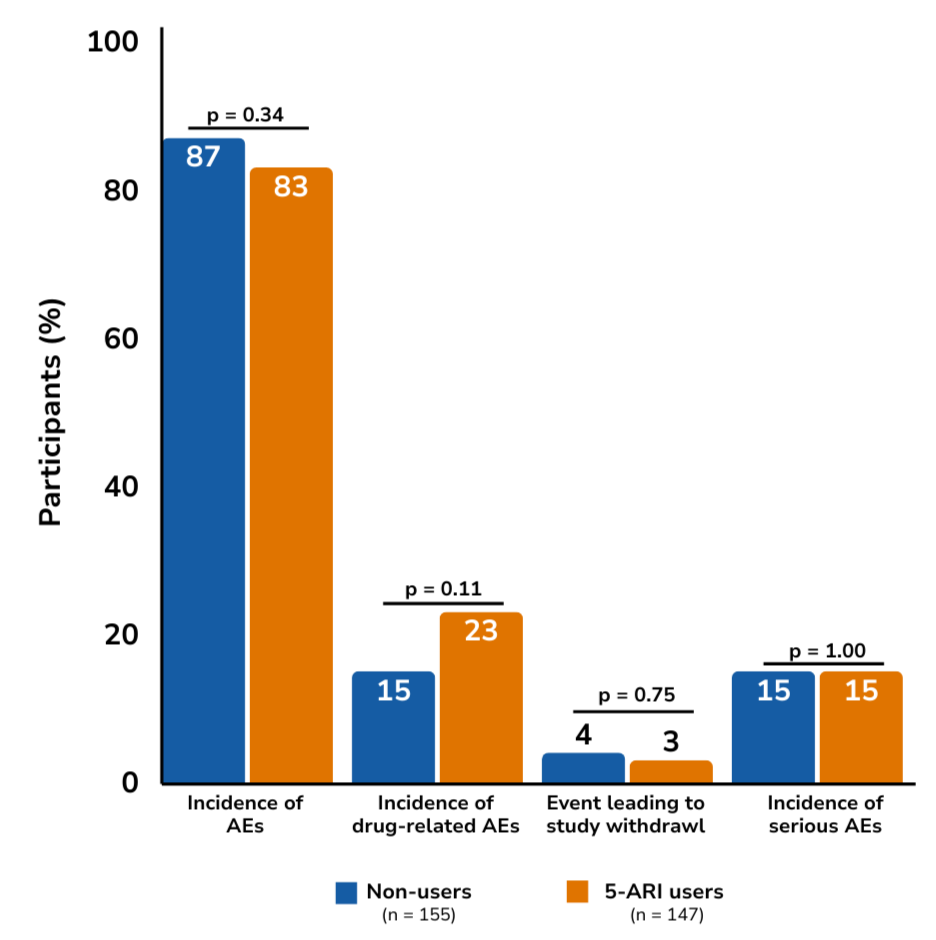
Adverse events
There were no significant differences in adverse events related to sexual function, breast disorders, or cardiovascular events. Sexual or breast-related adverse events occurred in 24% of patients receiving dutasteride, and 15% of patients receiving placebo, and cardiovascular events occurred in 5% of patients in both groups. No prostate cancer–related deaths or metastatic events were observed during the study period.
